AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Line spectra definition7/30/2023 
Therefore, the wavelength of the light emitted can help to determine which element has produced the light. The energy levels in the atom are unique to each element on the periodic table. The electrons that drop from the higher energy levels to the lower energy levels in an atom release a photon with a specific wavelength, which generates the atomic emission spectrum. Explain how the atomic emission spectra occur and how they relate to the elements on the periodic table.Īnswer. 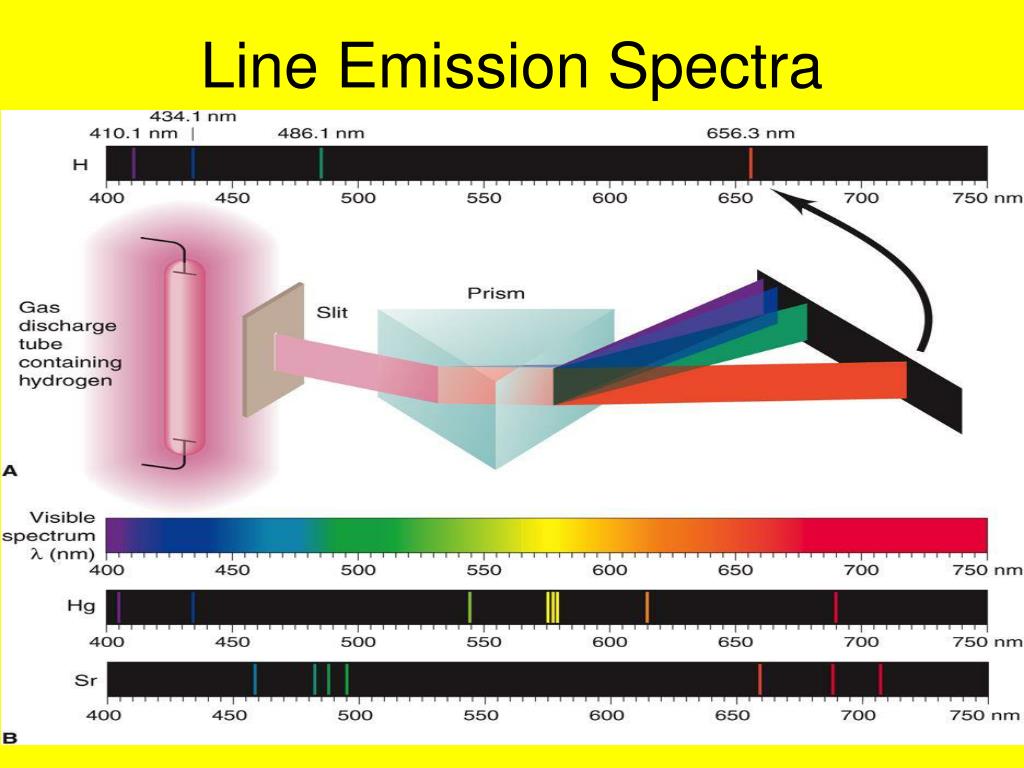
Atomic spectroscopy is also useful for occupational and environmental monitoring.It is applicable for identification of the spectral lines of materials used in metallurgy.It gives an accurate analytical method for finding components in a material that has an unknown chemical composition. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
